Stabilization of Different Types of Transition States in a Single Enzyme Active Site: QM/MM Analysis of Enzymes in the Alkaline Phosphatase Superfamily | Journal of the American Chemical Society
![Catalytic mechanism of the alkaline phosphatase reaction (see [34]). E,... | Download Scientific Diagram Catalytic mechanism of the alkaline phosphatase reaction (see [34]). E,... | Download Scientific Diagram](https://www.researchgate.net/publication/45288363/figure/fig1/AS:307398137925634@1450300835551/Catalytic-mechanism-of-the-alkaline-phosphatase-reaction-see-34-E-alkaline.png)
Catalytic mechanism of the alkaline phosphatase reaction (see [34]). E,... | Download Scientific Diagram

Phosphate Binding in the Active Site of Alkaline Phosphatase and the Interactions of 2-Nitrosoacetophenone with Alkaline Phosphatase-Induced Small Structural Changes: Biophysical Journal

Alkaline Phosphatase Catalysis Is Ultrasensitive to Charge Sequestered between the Active Site Zinc Ions | Journal of the American Chemical Society

Hydrolytic enzymes Zn(II) containing enzymes. Enzymatic catalysis of hydrolysis EnzymeMetal ion(s)Catalyzed reaction Alkaline phosphatase Purple acid. - ppt download

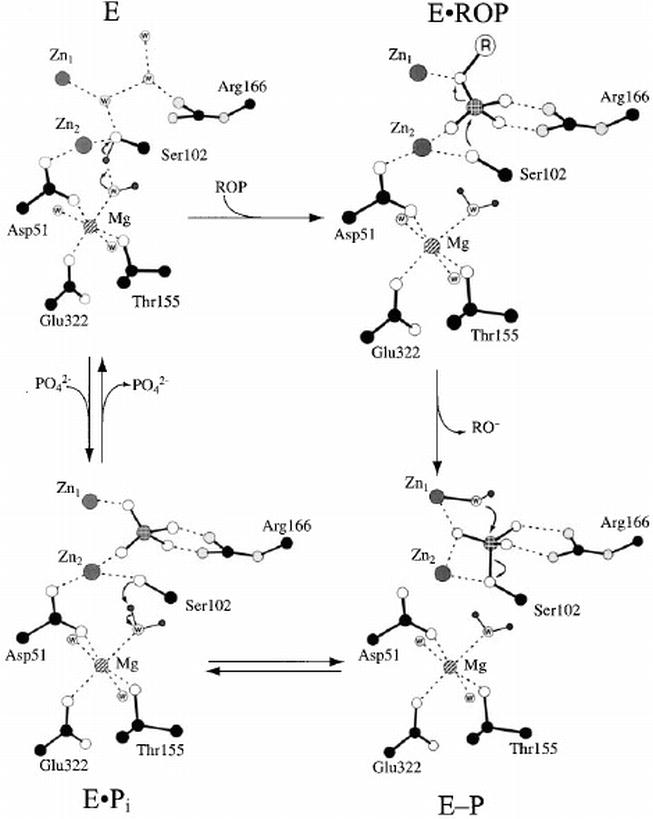
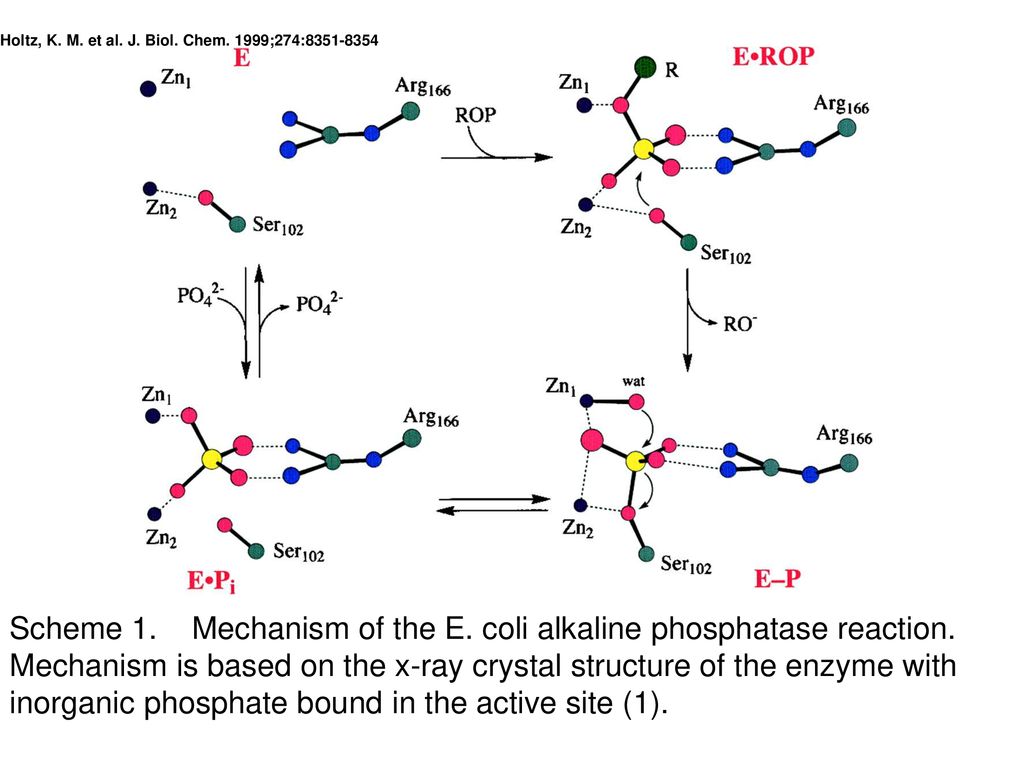
The mechanism of the alkaline phosphatase reaction: insights from NMR, crystallography and site‐specific mutagenesis - Holtz - 1999 - FEBS Letters - Wiley Online Library

A revised mechanism for the alkaline phosphatase reaction involving three metal ions - ScienceDirect

A new activity for an old enzyme: Escherichia coli bacterial alkaline phosphatase is a phosphite-dependent hydrogenase | PNAS

IJMS | Free Full-Text | Potent Alkaline Phosphatase Inhibitors, Pyrazolo-Oxothiazolidines: Synthesis, Biological Evaluation, Molecular Docking, and Kinetic Studies
![PDF] Comparative enzymology in the alkaline phosphatase superfamily to determine the catalytic role of an active-site metal ion. | Semantic Scholar PDF] Comparative enzymology in the alkaline phosphatase superfamily to determine the catalytic role of an active-site metal ion. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c2805cbf0a7441ef30ae3f36d23792c5cabf4e11/2-Figure1-1.png)
PDF] Comparative enzymology in the alkaline phosphatase superfamily to determine the catalytic role of an active-site metal ion. | Semantic Scholar
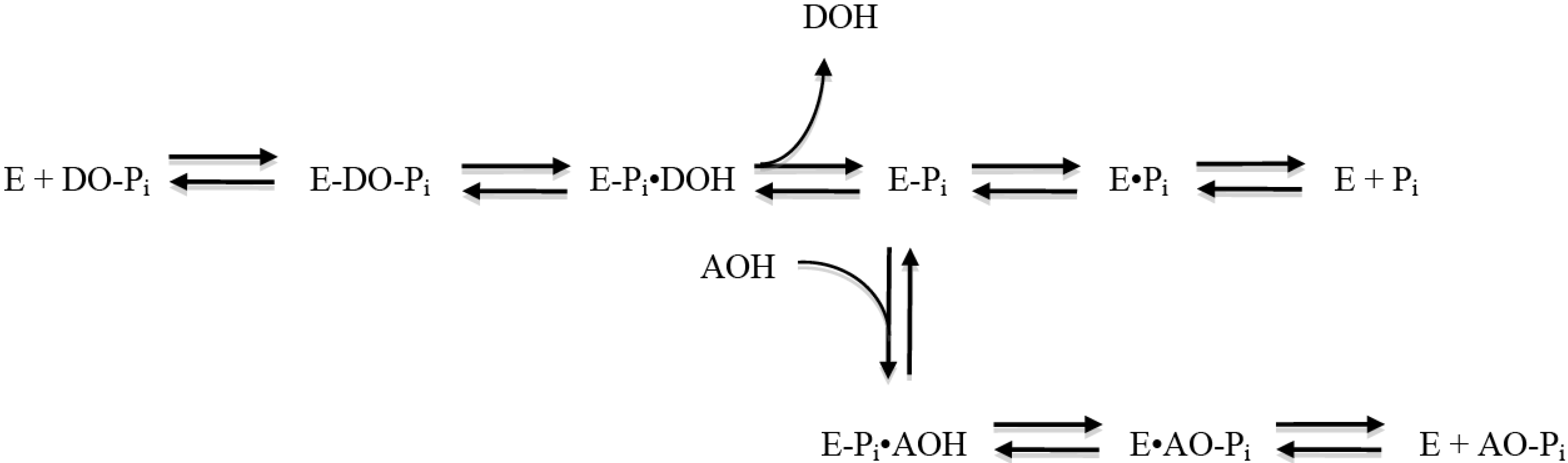
Molecules | Free Full-Text | Assay Format as a Critical Success Factor for Identification of Novel Inhibitor Chemotypes of Tissue-Nonspecific Alkaline Phosphatase from High-Throughput Screening
X-Ray Structure Reveals a New Class and Provides Insight into Evolution of Alkaline Phosphatases | PLOS ONE