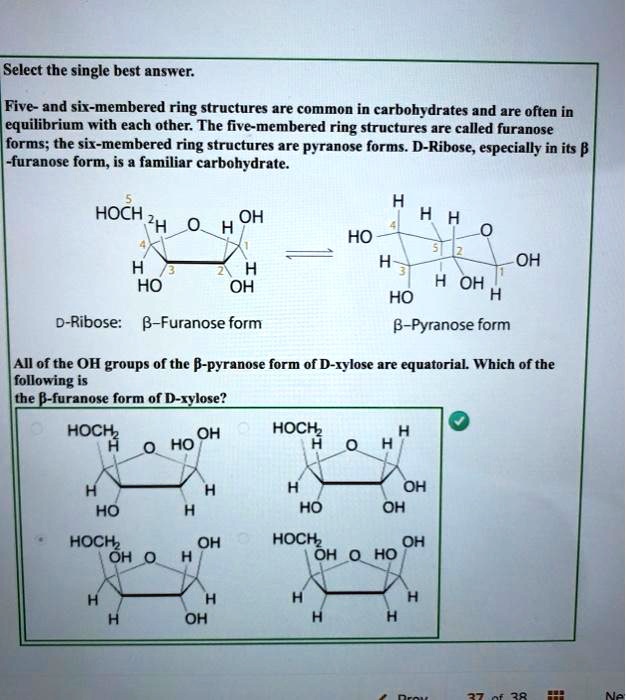
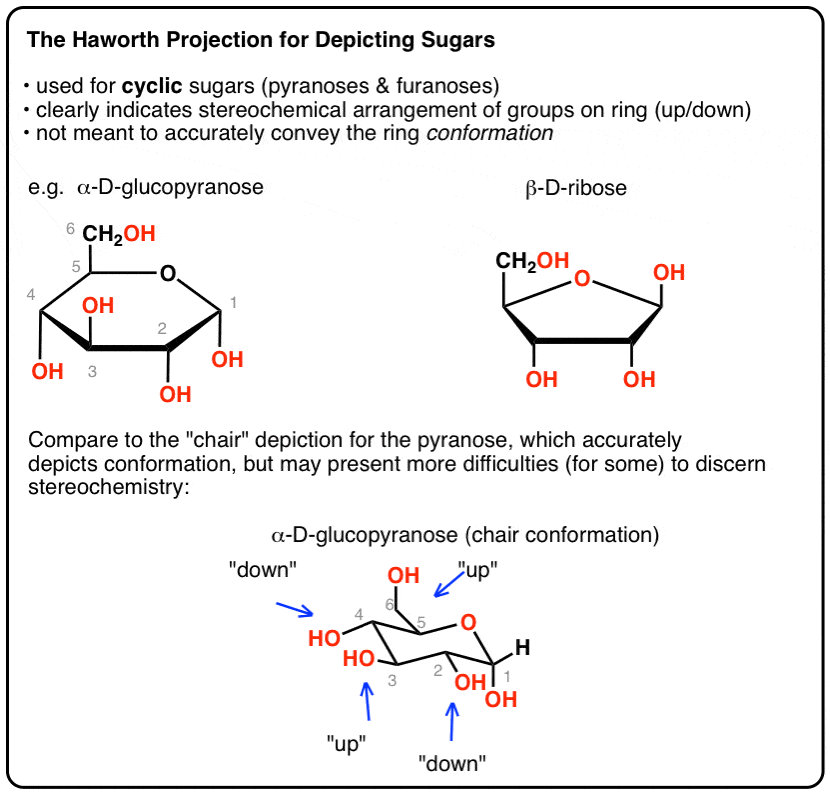
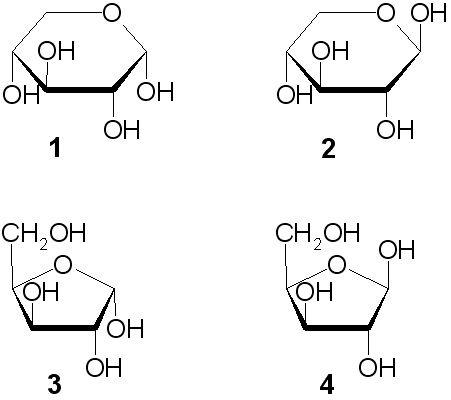
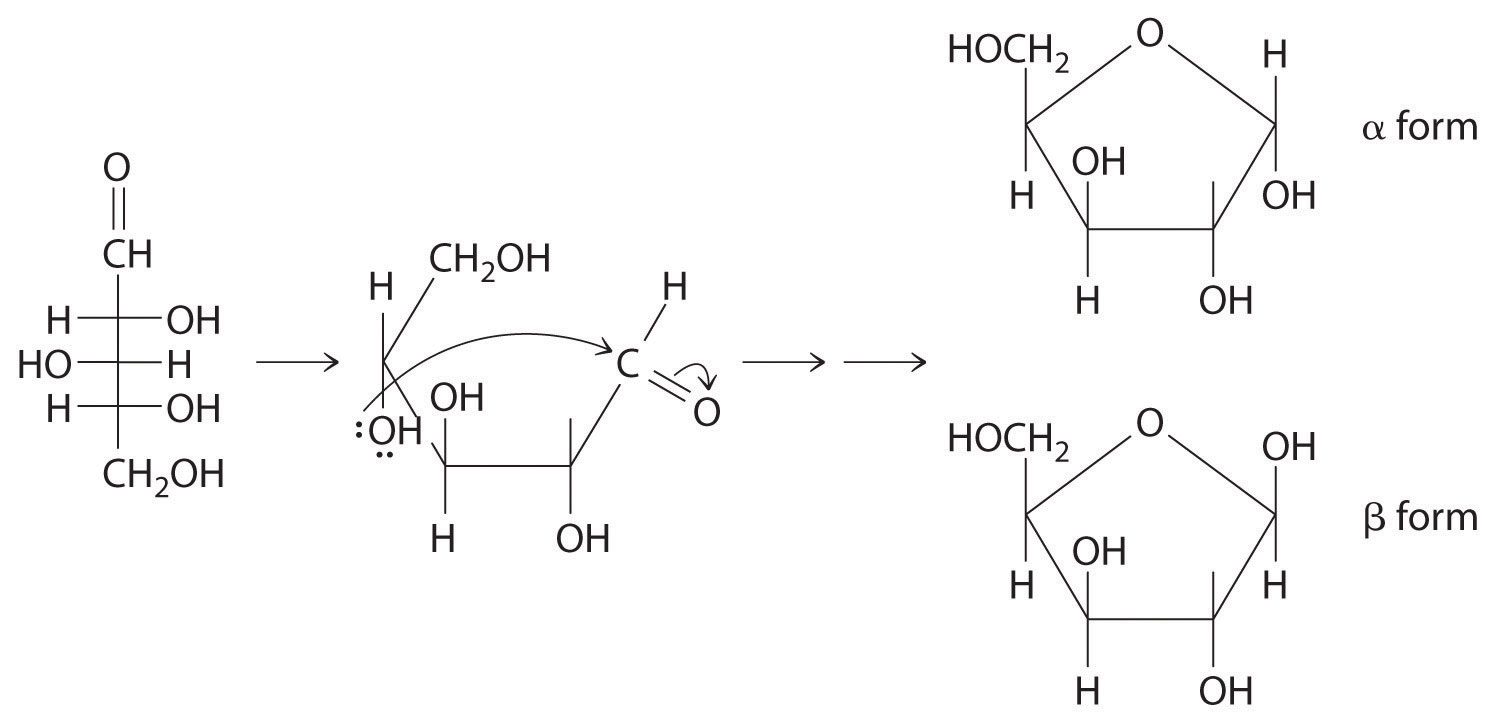
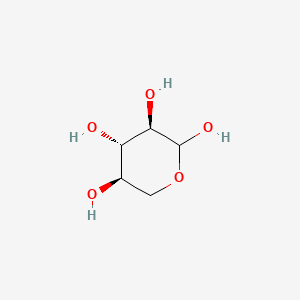
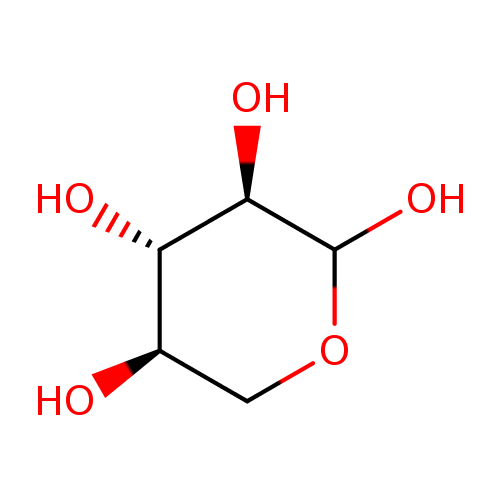
Scheme 1 Fisher projection of D-xylose (centre) and Haworth projections... | Download Scientific Diagram

Hemicellulose is a plant polysaccharide. It is a heteropolymer, meaning it contains more than one monosaccharide. D-xylose residues are present in the greatest amount and are attached by (1->4) glycosidic bonds. Draw

Unraveling substituent effects on the glass transition temperatures of biorenewable polyesters | Nature Communications

Polymers from Sugars and Cyclic Anhydrides: Ring-Opening Copolymerization of a d-Xylose Anhydrosugar Oxetane | Macromolecules