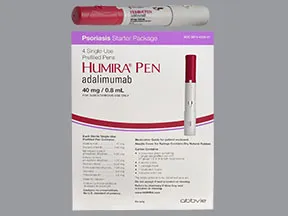
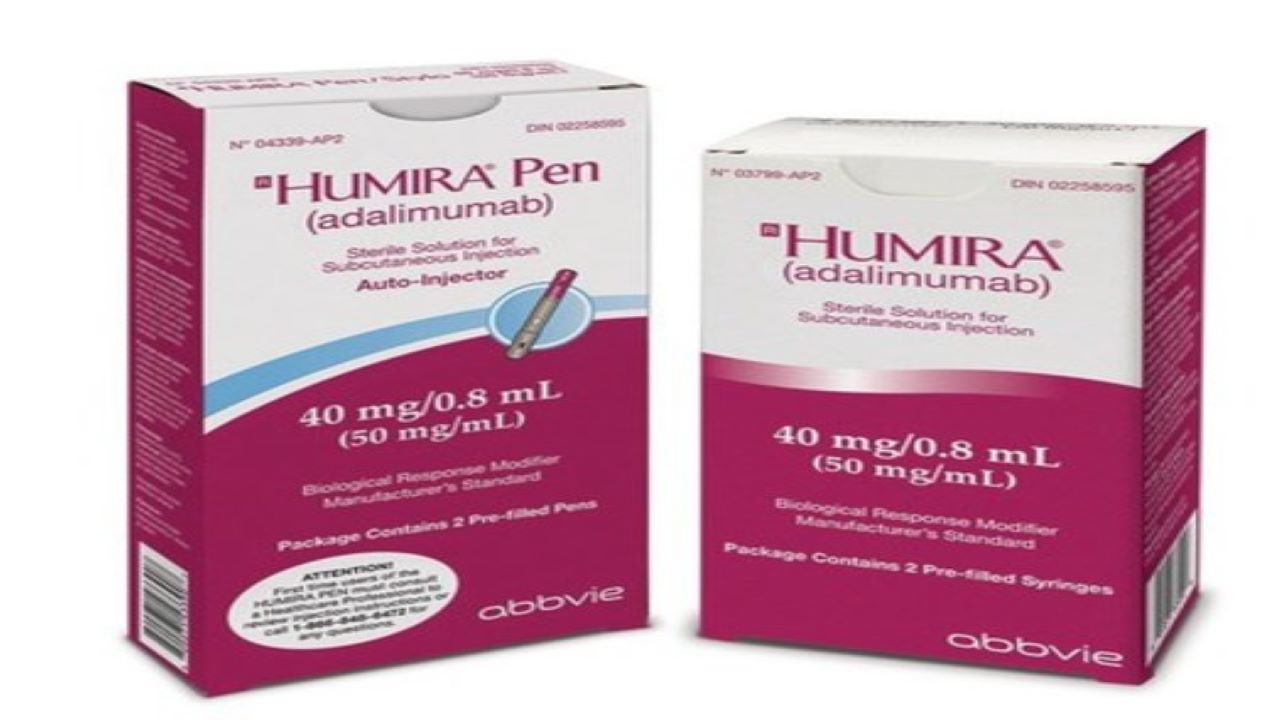
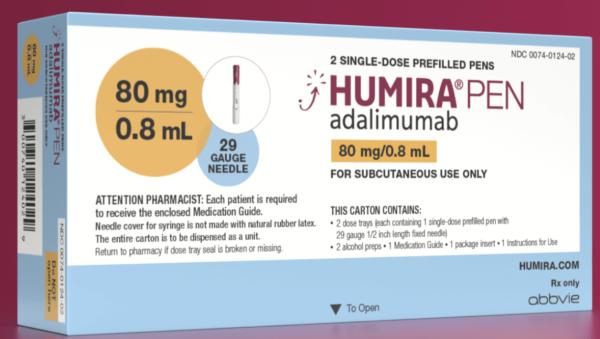
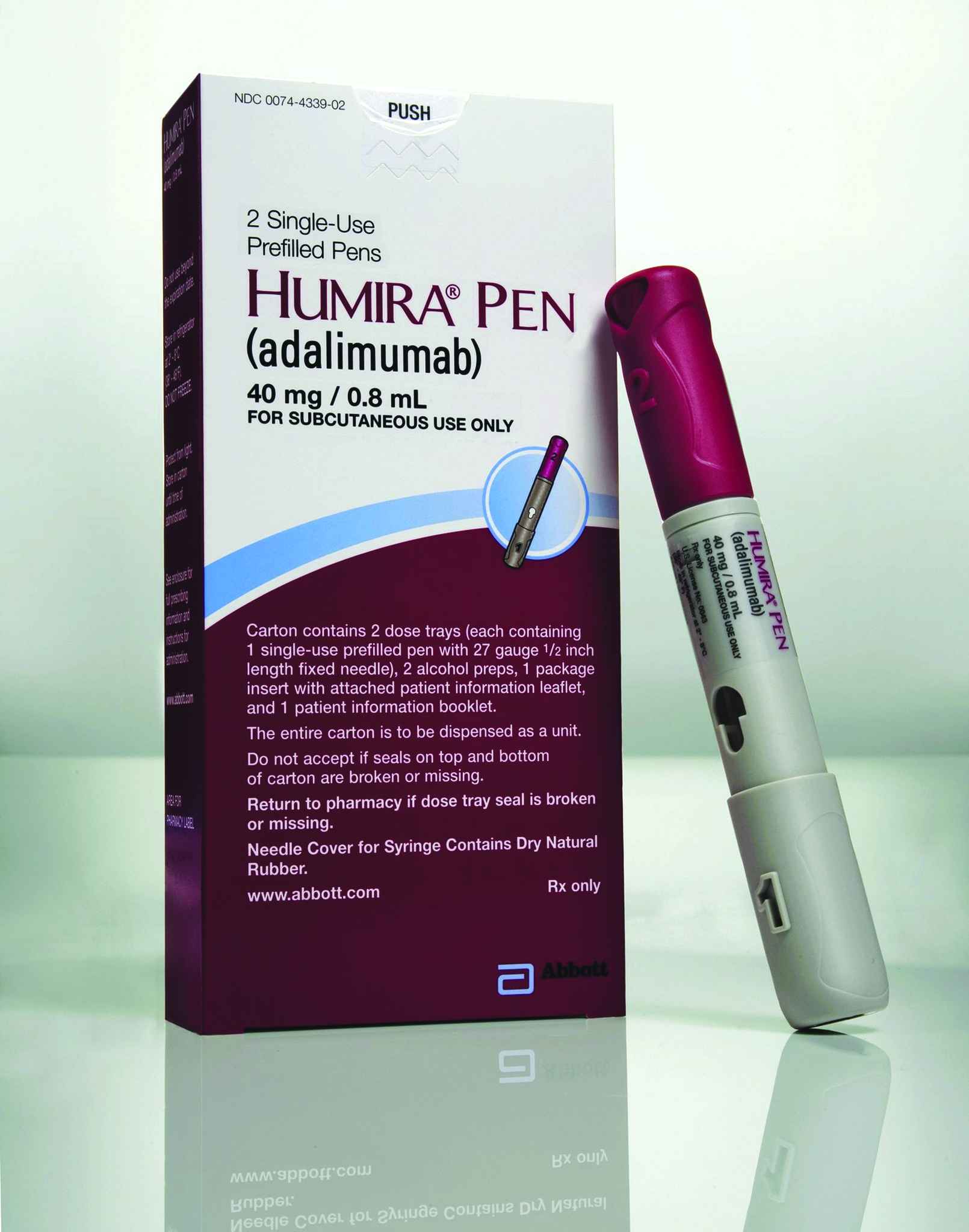
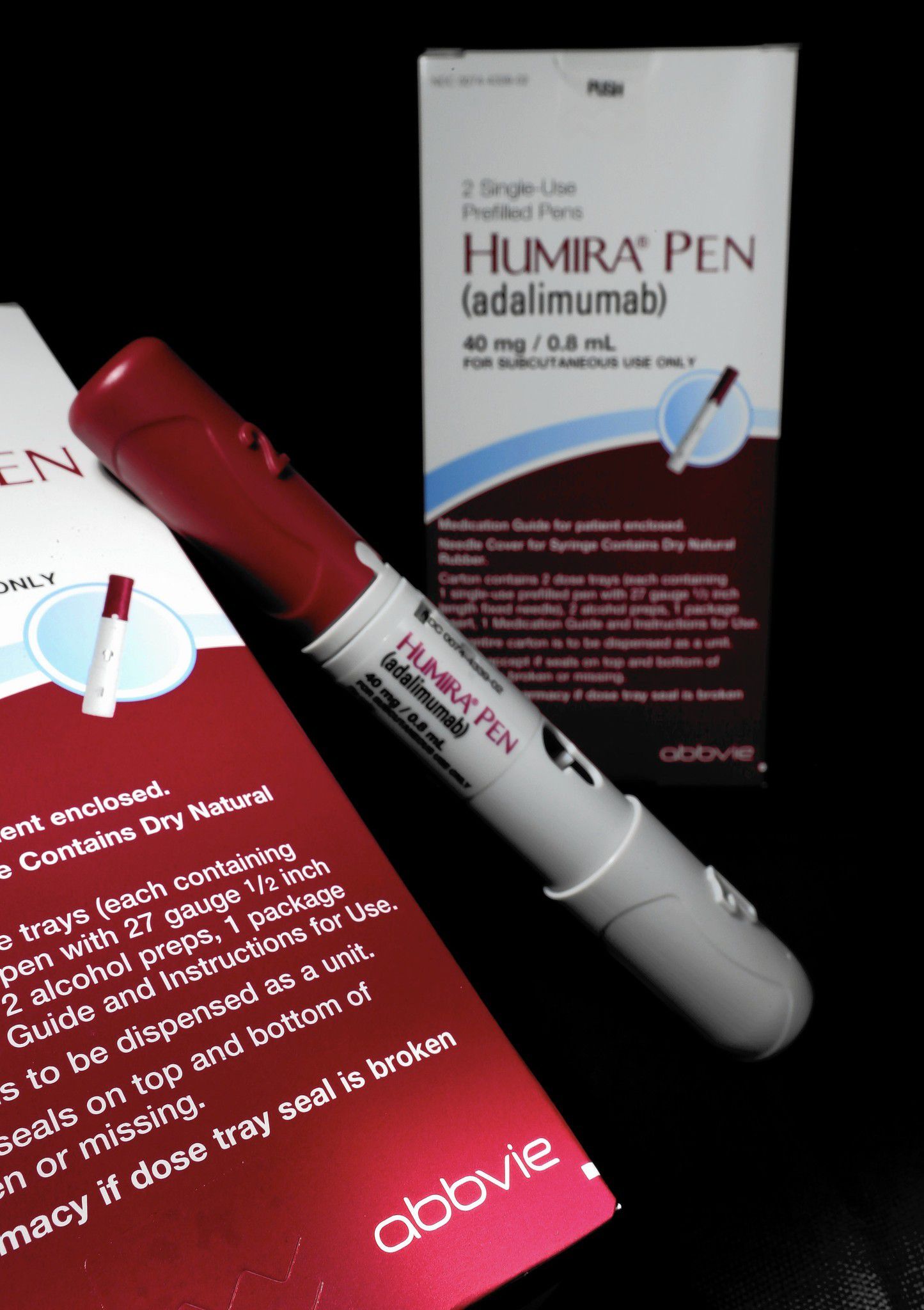
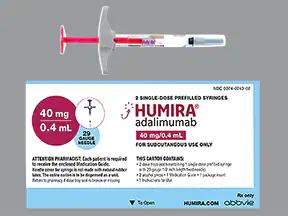
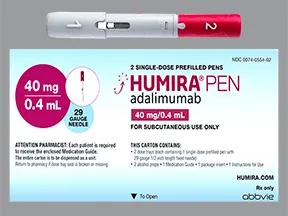
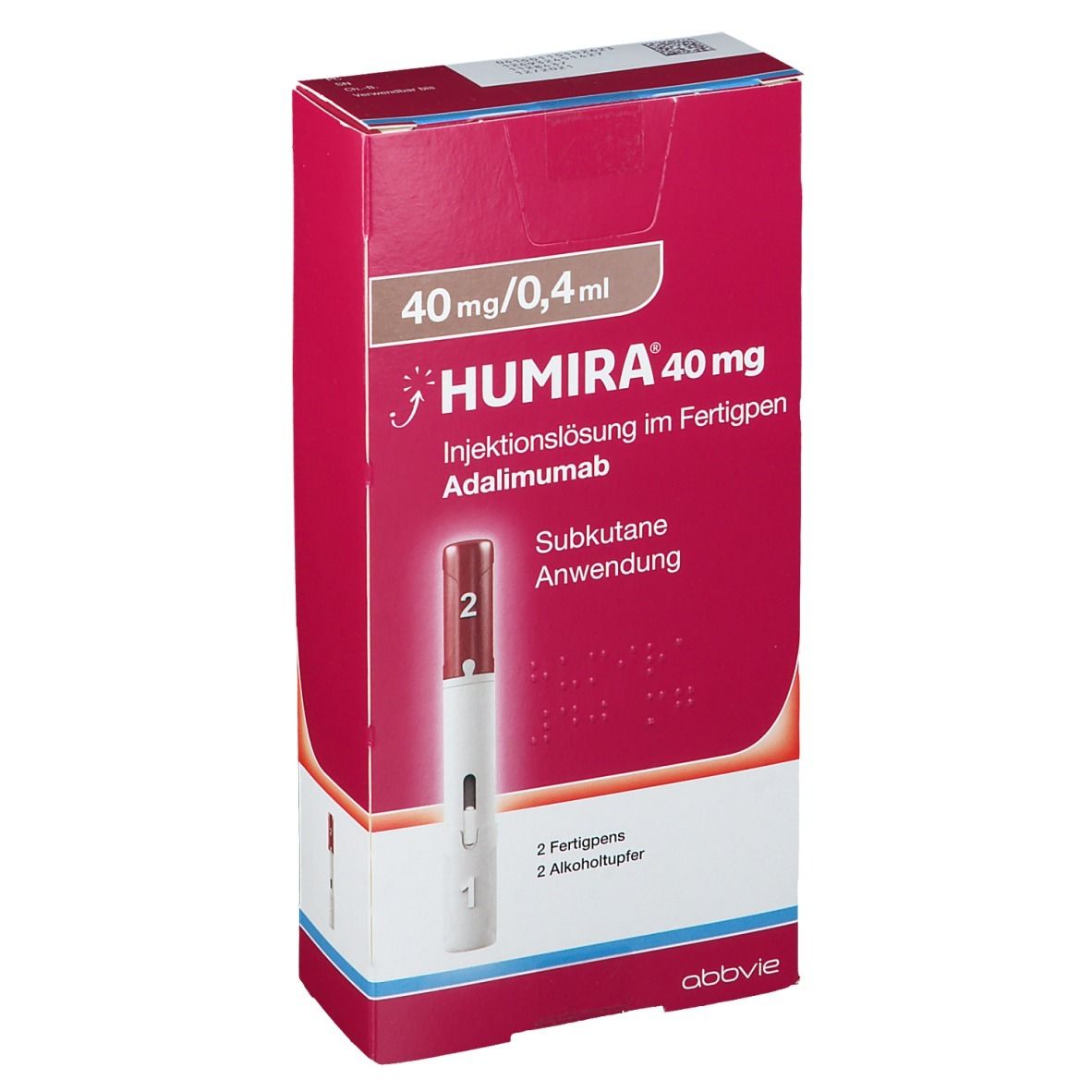
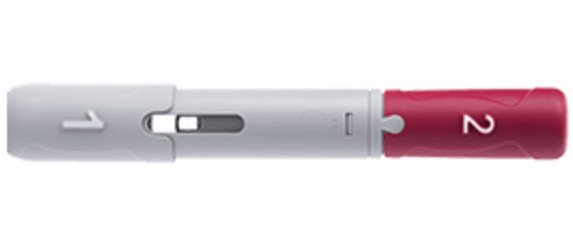
Humira Crohn's Disease Starter Pack Disease-Modifying Antirheumatic Agent Adalimumab, Preservative Free 40 mg / 0.8 mL Subcutaneous Injection Prefilled Auto-Injector Pen 6 Doses - Suprememed
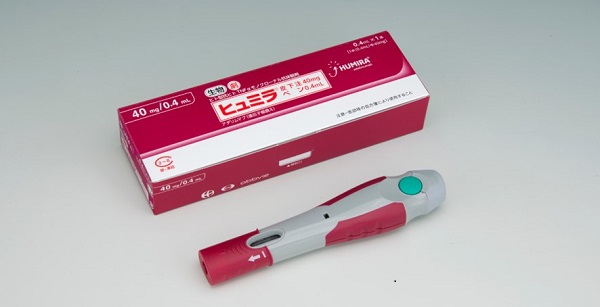
AbbVie and Eisai Announce the Launch of HUMIRA® Pen, an Auto-Injector Formulation for Fully Human Anti-TNF-α Monoclonal Antibody HUMIRA® | News Release:2018 | Eisai Co., Ltd.

AbbVie, Humira among products, companies facing penalty under drug pricing rule | Crain's Chicago Business