Understanding the Mechanism of B12-Dependent Methylmalonyl-CoA Mutase: Partial Proton Transfer in Action | Journal of the American Chemical Society
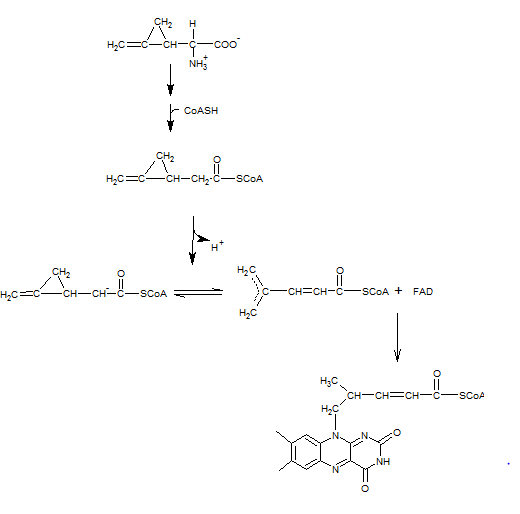
Based on the mechanism for the methylmalonyl CoA mutase, a reasonable mechanism for the given reactions should be written. Concept Introduction: In the methylmalonyl CoA mutase reaction, a free radical is formed
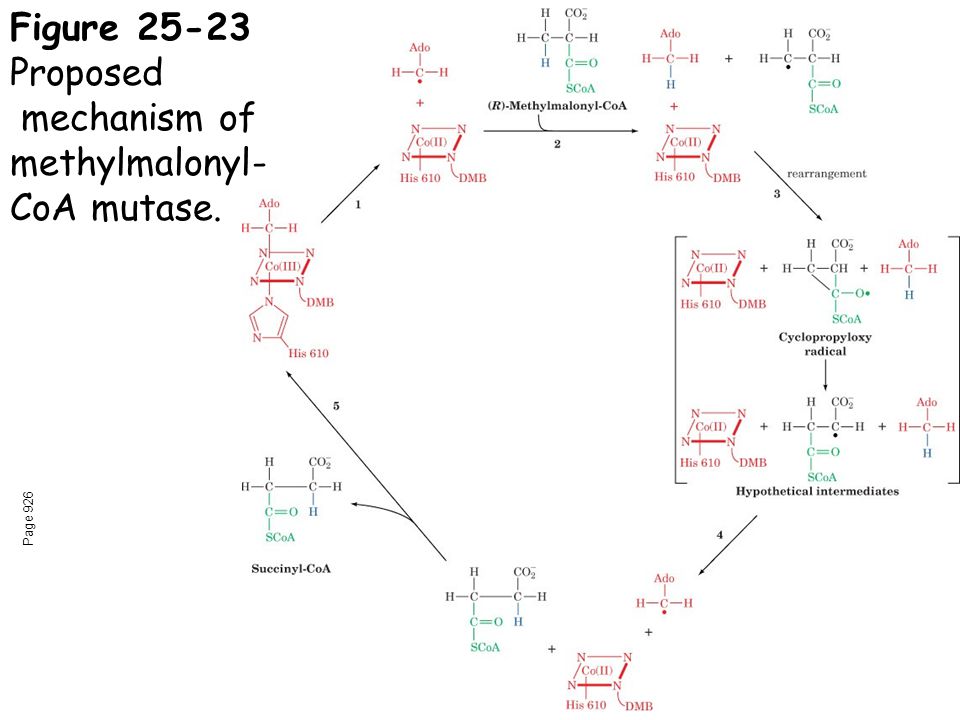
Figure 24-17Schematic representation of the thylakoid membrane showing the components of its electron-transport chain. Page ppt download

The Coenzyme B12 Analog 5′-Deoxyadenosylcobinamide-GDP Supports Catalysis by Methylmalonyl-CoA Mutase in the Absence of Trans-ligand Coordination* - Journal of Biological Chemistry
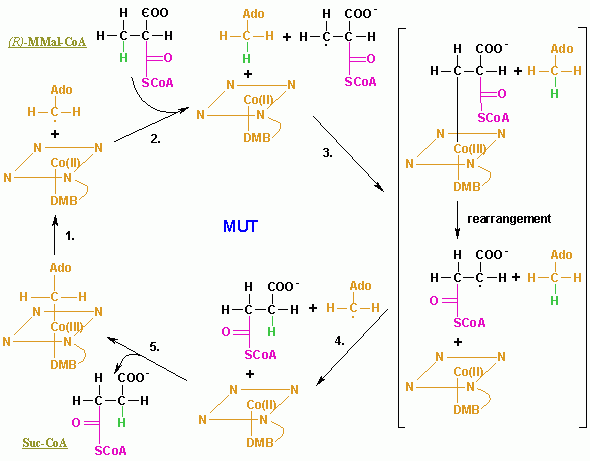
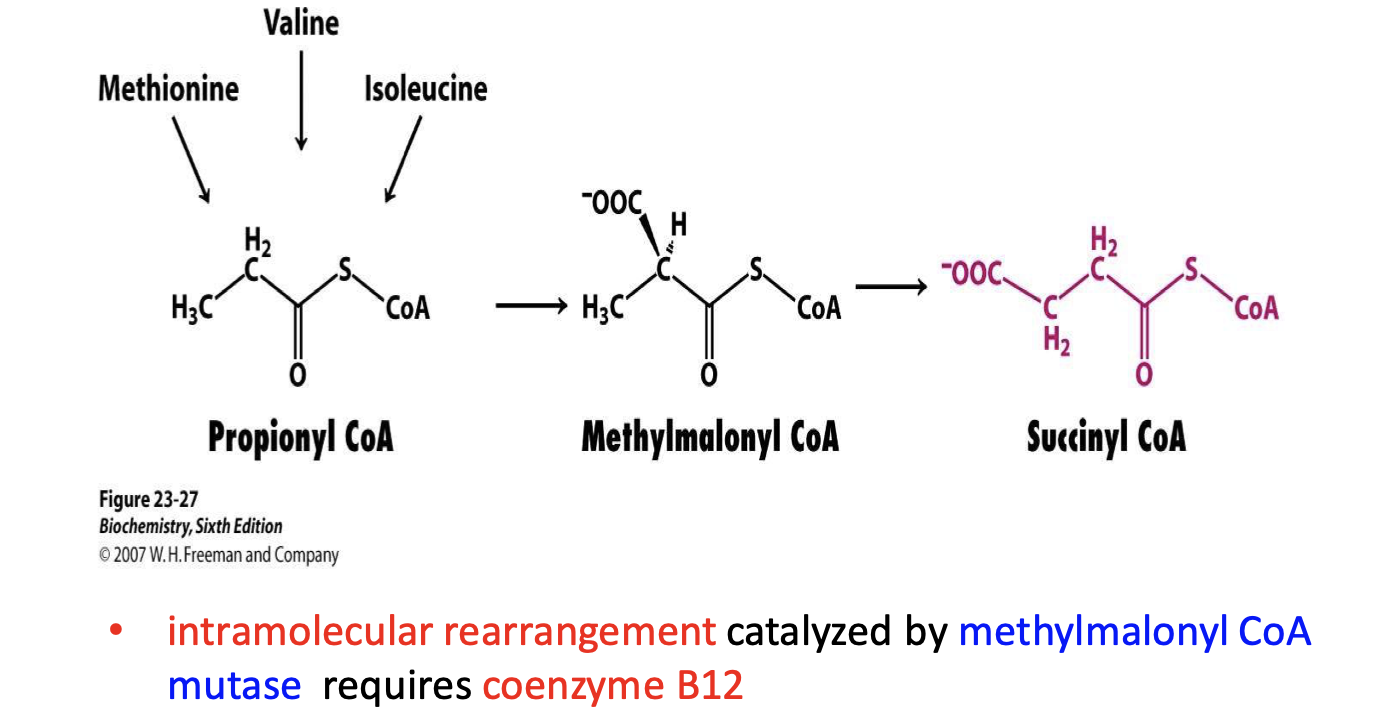
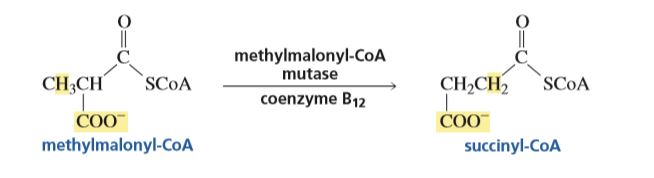
Methylmalonyl-CoA mutase: Conversion of (R)-methylmalonyl-CoA to succinyl- CoA (degradation of odd- carbon fatty acids)

Figure 4 from Crystal structure of substrate complexes of methylmalonyl-CoA mutase. | Semantic Scholar
Methylmalonyl-CoA mutase: Conversion of (R)-methylmalonyl-CoA to succinyl- CoA (degradation of odd- carbon fatty acids)

Figure 1 from Methylmalonyl-coA epimerase deficiency: A new case, with an acute metabolic presentation and an intronic splicing mutation in the MCEE gene | Semantic Scholar

Naturally occurring cobalamin (B12) analogs can function as cofactors for human methylmalonyl-CoA mutase | bioRxiv
Methylmalonyl-CoA mutase: Conversion of (R)-methylmalonyl-CoA to succinyl- CoA (degradation of odd- carbon fatty acids)

Methylmalonyl-CoA Mutase Induction by Cerebral Ischemia and Neurotoxicity of the Mitochondrial Toxin Methylmalonic Acid | Journal of Neuroscience

Postulated reaction mechanism of methylmalonyl-CoA mutase. MCoA and... | Download Scientific Diagram

Understanding the Mechanism of B12-Dependent Methylmalonyl-CoA Mutase: Partial Proton Transfer in Action | Journal of the American Chemical Society