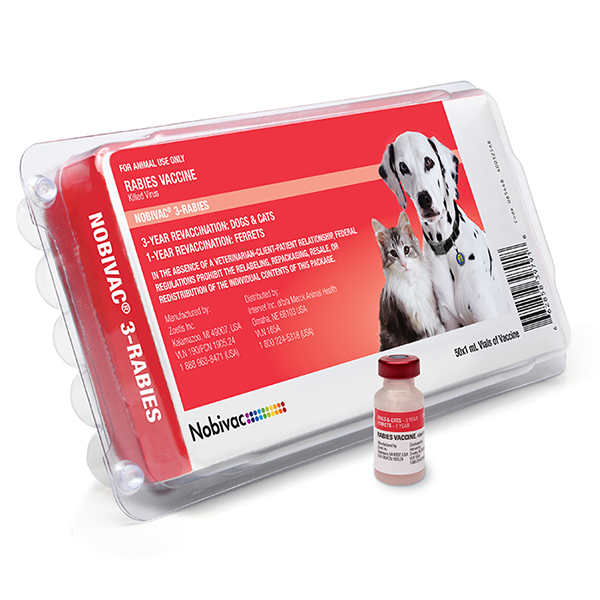
📅 Dog Rabies Vaccine: Recommended Timeline 📅 💉 1 Year Rabies Vaccine 💉 Safe to Start: 12 Weeks Booster: 1 Year from Date Given 💉 3 Year Rabies Vaccine... | By University Drive Veterinary Hospital | Facebook

Use of a Modified Preexposure Prophylaxis Vaccination Schedule to Prevent Human Rabies: Recommendations of the Advisory Committee on Immunization Practices — United States, 2022 | MMWR
Persistence of Rabies Virus-Neutralizing Antibodies after Vaccination of Rural Population following Vampire Bat Rabies Outbreak in Brazil | PLOS Neglected Tropical Diseases

Reduced dose pre-exposure primary and booster intradermal rabies vaccination with a purified chick embryo cell vaccine (PCECV) is immunogenic and safe in adults. | Semantic Scholar

Safety and immunogenicity of a mRNA rabies vaccine in healthy adults: an open-label, non-randomised, prospective, first-in-human phase 1 clinical trial - The Lancet

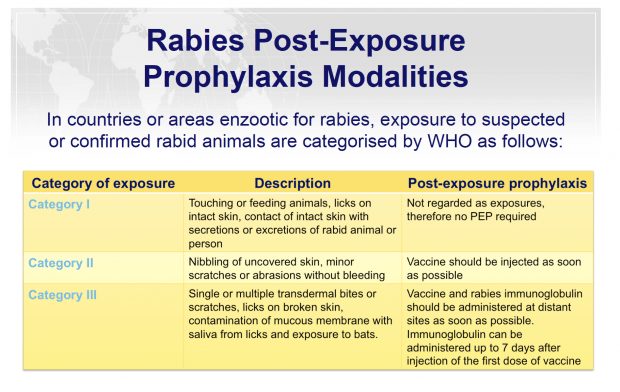
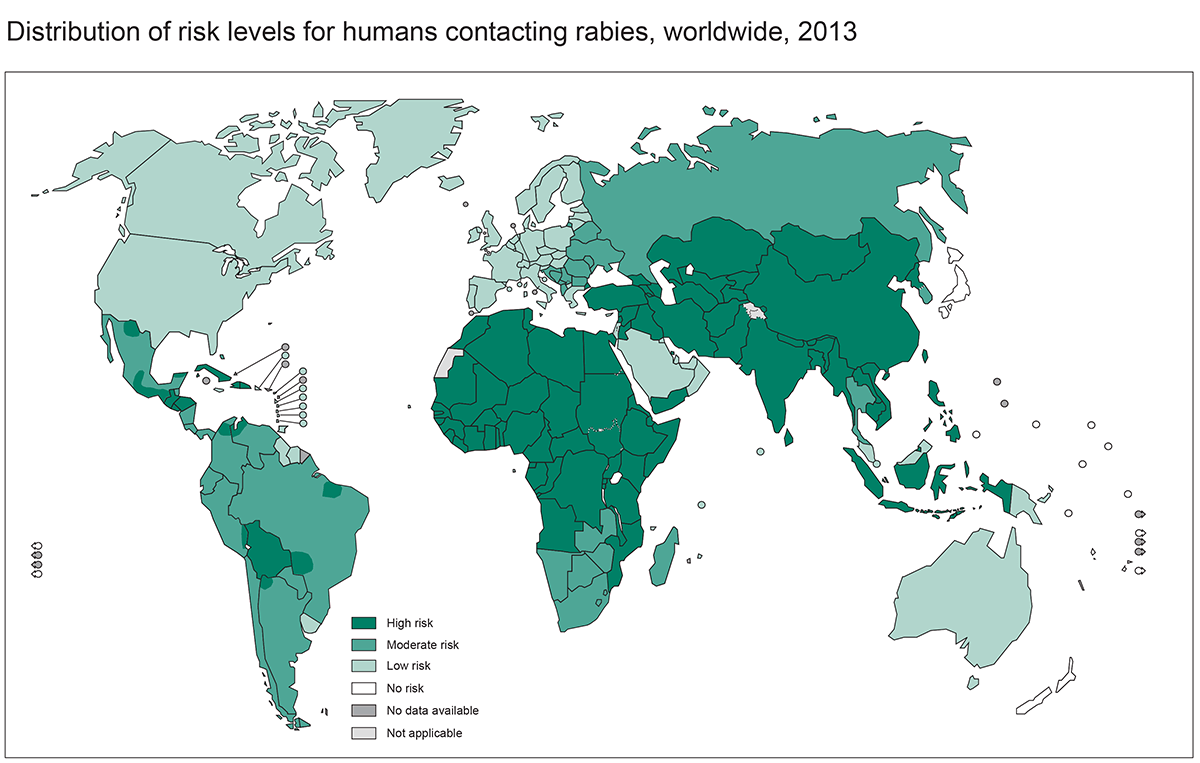
Figure. Rabies post-exposure prophylaxis: terrestrial animal exposures | The Australian Immunisation Handbook





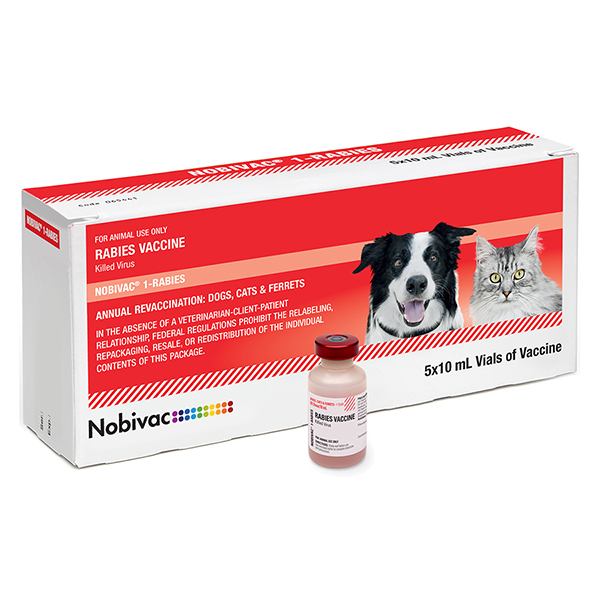
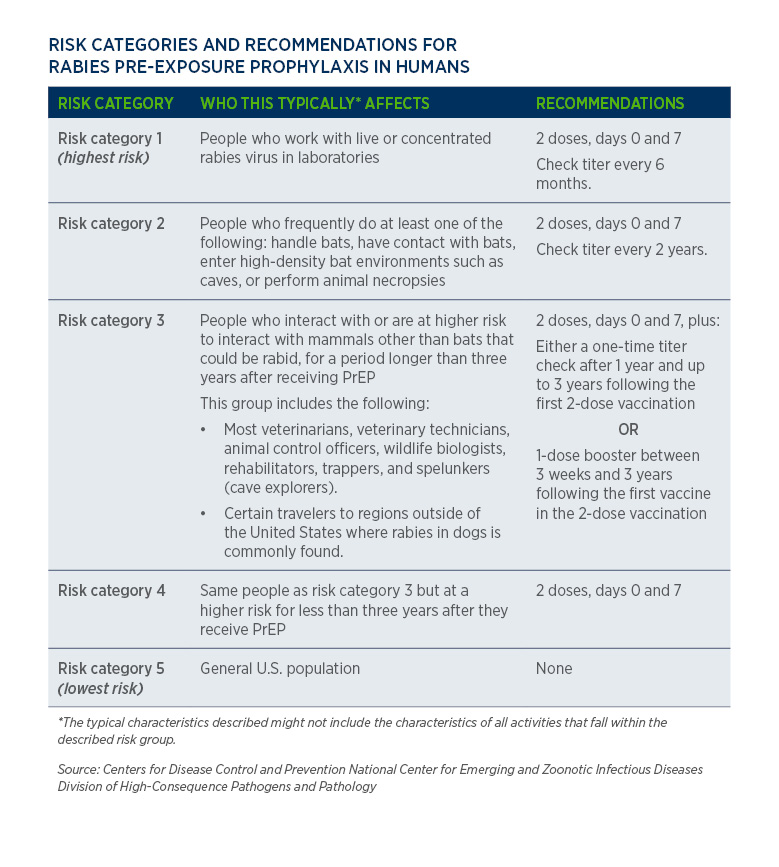
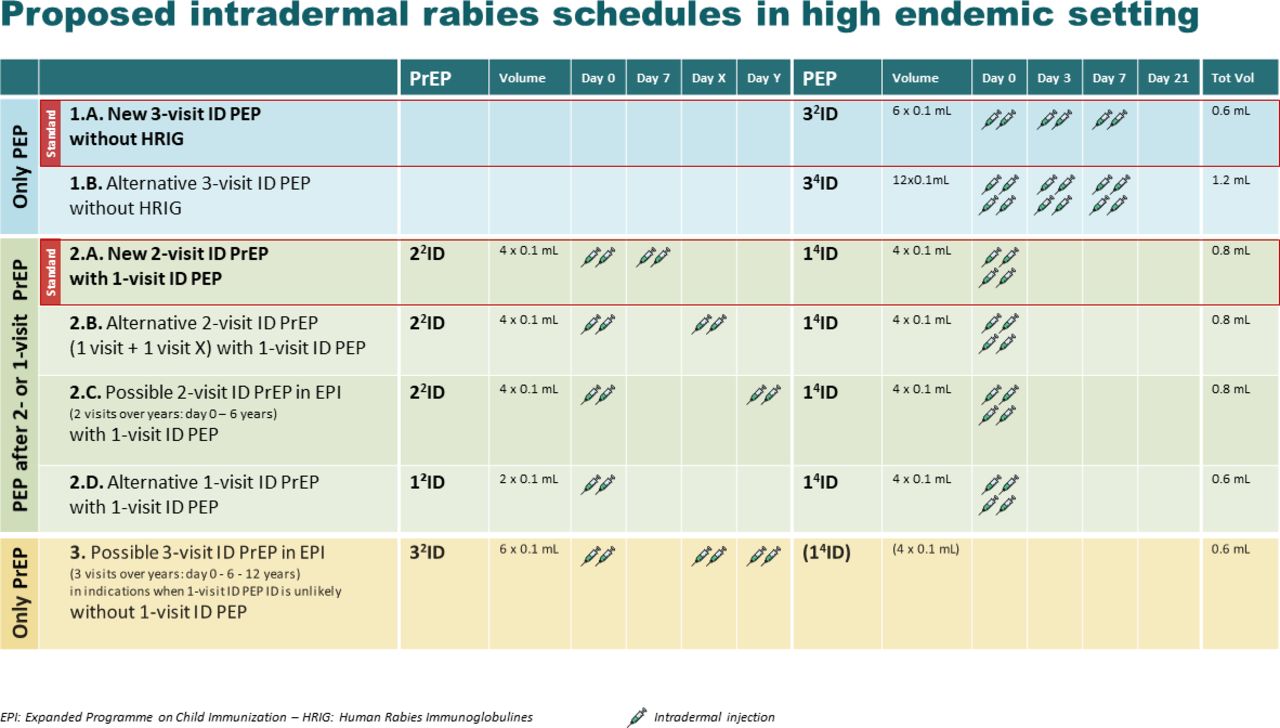
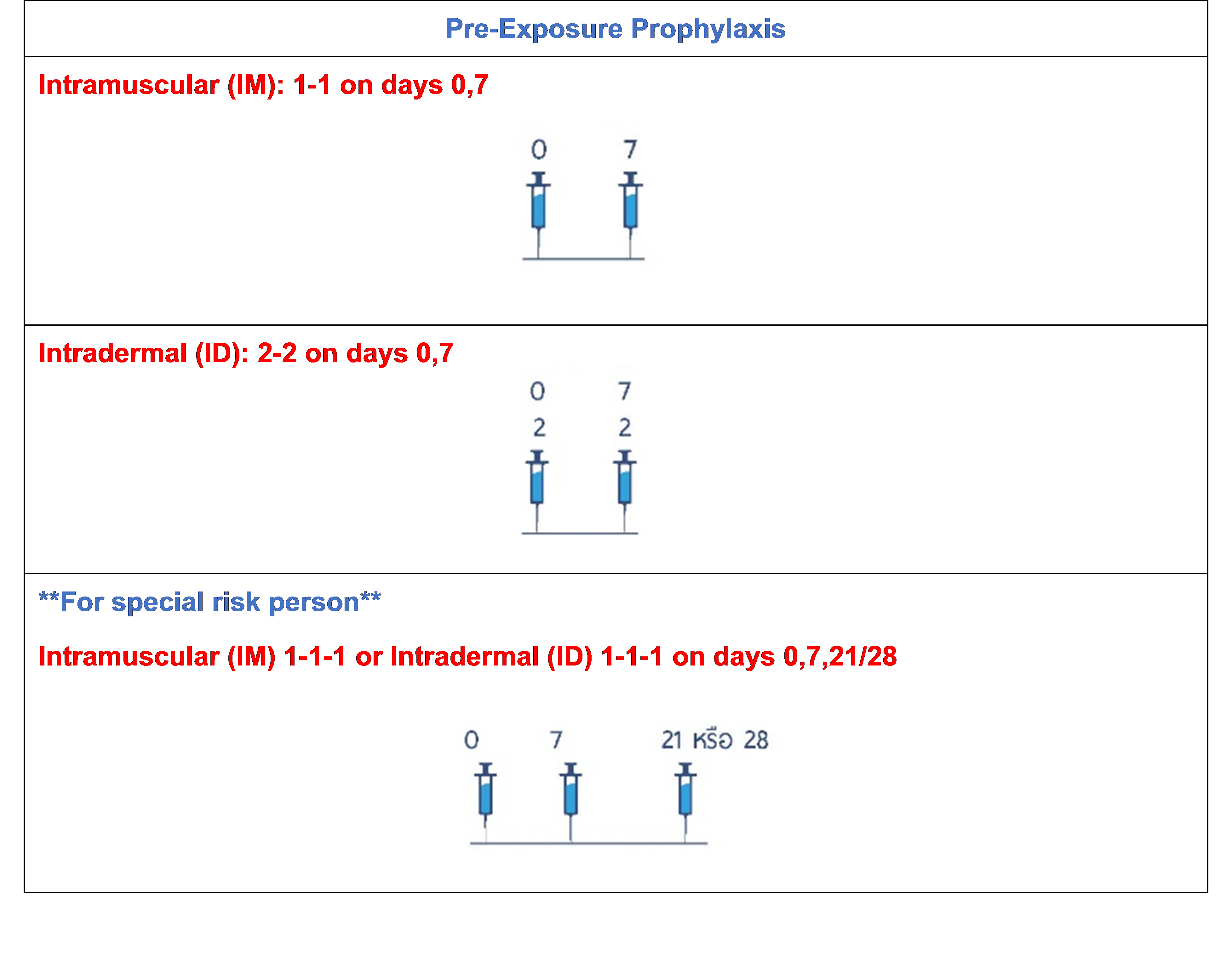
.jpg)
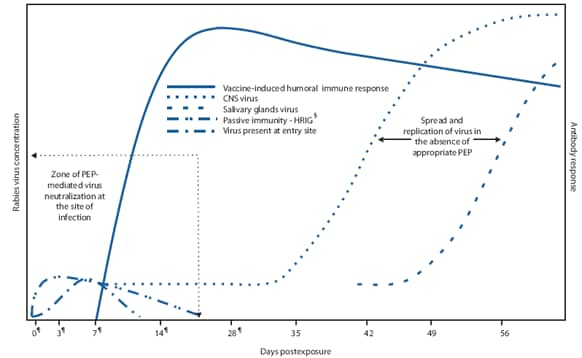

:strip_icc()/how-long-are-rabies-shots-good-3385625_FINAL-b3b90b78675e4a9ca4102b405fecf74c.jpg)